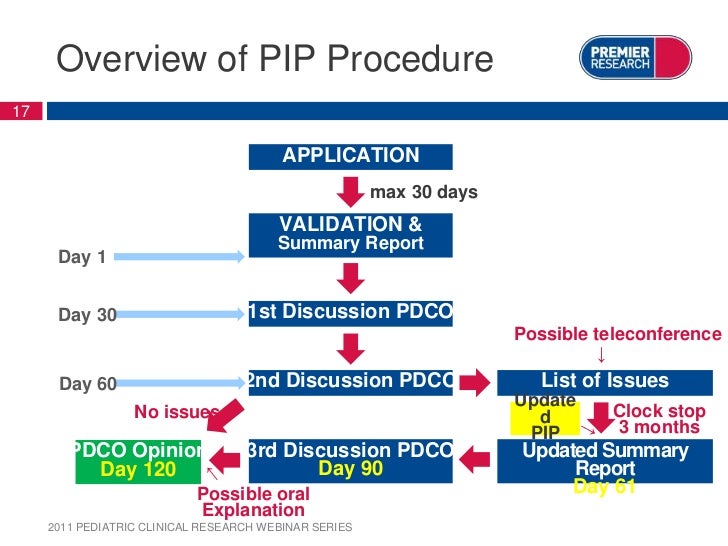
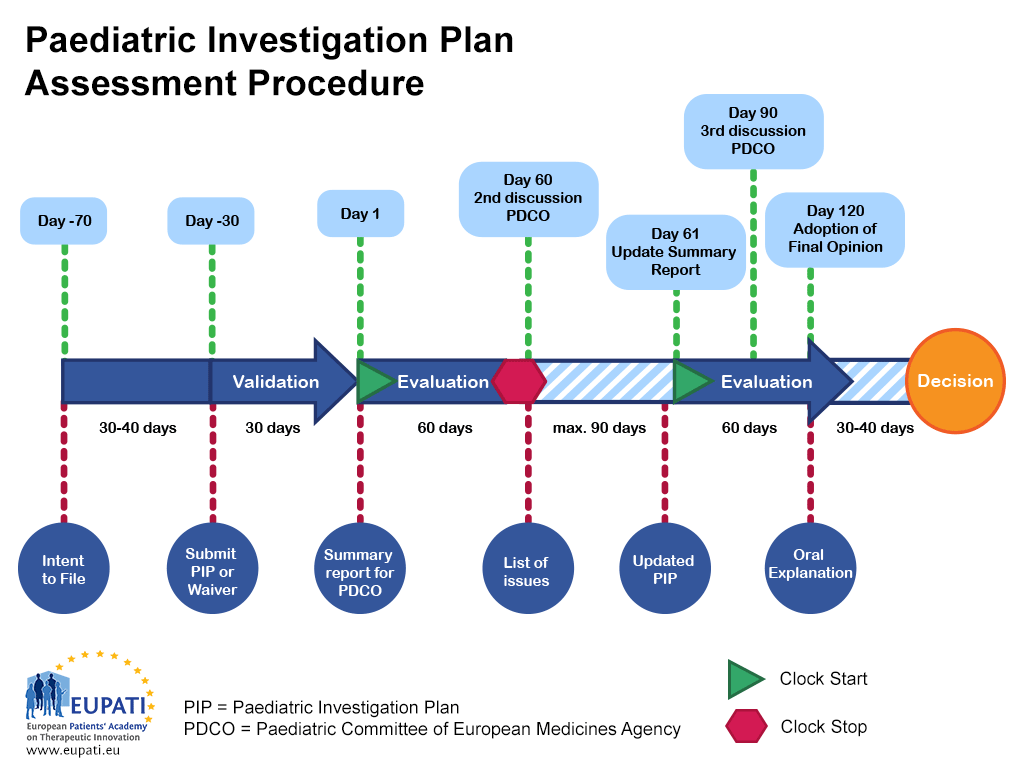
Paediatric Investigation Plan Template
Paediatric Investigation Plan Template - Web submission of a paediatric investigation plan must be made to the mhra no later than the completion of the human pharmaco. Content of and process for submitting initial pediatric study plans and amended initial pediatric study. Web this page lists the templates and forms required by companies wishing to apply for a paediatric investigation plan (pip), deferral. Web the main challenges for medical writers when writing a pip are application of the guidance to the drug and disease in hand,.
Content of and process for submitting initial pediatric study plans and amended initial pediatric study. Web submission of a paediatric investigation plan must be made to the mhra no later than the completion of the human pharmaco. Web this page lists the templates and forms required by companies wishing to apply for a paediatric investigation plan (pip), deferral. Web the main challenges for medical writers when writing a pip are application of the guidance to the drug and disease in hand,.
Web the main challenges for medical writers when writing a pip are application of the guidance to the drug and disease in hand,. Content of and process for submitting initial pediatric study plans and amended initial pediatric study. Web this page lists the templates and forms required by companies wishing to apply for a paediatric investigation plan (pip), deferral. Web submission of a paediatric investigation plan must be made to the mhra no later than the completion of the human pharmaco.
Pediatric Investigation Plans Part 1 Determining Taste Masking
Web submission of a paediatric investigation plan must be made to the mhra no later than the completion of the human pharmaco. Content of and process for submitting initial pediatric study plans and amended initial pediatric study. Web the main challenges for medical writers when writing a pip are application of the guidance to the drug and disease in hand,..
Outsourcing Paediatric Investigation Plans
Web this page lists the templates and forms required by companies wishing to apply for a paediatric investigation plan (pip), deferral. Web the main challenges for medical writers when writing a pip are application of the guidance to the drug and disease in hand,. Web submission of a paediatric investigation plan must be made to the mhra no later than.
Planning your Paediatric Investigation Plan (PIP) Submission in Euro…
Web submission of a paediatric investigation plan must be made to the mhra no later than the completion of the human pharmaco. Web the main challenges for medical writers when writing a pip are application of the guidance to the drug and disease in hand,. Web this page lists the templates and forms required by companies wishing to apply for.
Planning your Paediatric Investigation Plan (PIP) Submission in Euro…
Web the main challenges for medical writers when writing a pip are application of the guidance to the drug and disease in hand,. Web this page lists the templates and forms required by companies wishing to apply for a paediatric investigation plan (pip), deferral. Web submission of a paediatric investigation plan must be made to the mhra no later than.
Planning your Paediatric Investigation Plan (PIP) Submission in Euro…
Content of and process for submitting initial pediatric study plans and amended initial pediatric study. Web this page lists the templates and forms required by companies wishing to apply for a paediatric investigation plan (pip), deferral. Web submission of a paediatric investigation plan must be made to the mhra no later than the completion of the human pharmaco. Web the.
Overview of current paediatric investigation plan (PIP) application
Web the main challenges for medical writers when writing a pip are application of the guidance to the drug and disease in hand,. Web submission of a paediatric investigation plan must be made to the mhra no later than the completion of the human pharmaco. Content of and process for submitting initial pediatric study plans and amended initial pediatric study..
Planning your Paediatric Investigation Plan (PIP) Submission in Euro…
Web the main challenges for medical writers when writing a pip are application of the guidance to the drug and disease in hand,. Web this page lists the templates and forms required by companies wishing to apply for a paediatric investigation plan (pip), deferral. Web submission of a paediatric investigation plan must be made to the mhra no later than.
Planning your Paediatric Investigation Plan (PIP) Submission in Euro…
Web submission of a paediatric investigation plan must be made to the mhra no later than the completion of the human pharmaco. Web this page lists the templates and forms required by companies wishing to apply for a paediatric investigation plan (pip), deferral. Content of and process for submitting initial pediatric study plans and amended initial pediatric study. Web the.
Paediatric medicine Paediatric Investigation Plan EUPATI Toolbox
Content of and process for submitting initial pediatric study plans and amended initial pediatric study. Web submission of a paediatric investigation plan must be made to the mhra no later than the completion of the human pharmaco. Web this page lists the templates and forms required by companies wishing to apply for a paediatric investigation plan (pip), deferral. Web the.
Pediatric Care Plan Template Medical Diagnosis Nursing
Content of and process for submitting initial pediatric study plans and amended initial pediatric study. Web this page lists the templates and forms required by companies wishing to apply for a paediatric investigation plan (pip), deferral. Web submission of a paediatric investigation plan must be made to the mhra no later than the completion of the human pharmaco. Web the.
Content Of And Process For Submitting Initial Pediatric Study Plans And Amended Initial Pediatric Study.
Web the main challenges for medical writers when writing a pip are application of the guidance to the drug and disease in hand,. Web this page lists the templates and forms required by companies wishing to apply for a paediatric investigation plan (pip), deferral. Web submission of a paediatric investigation plan must be made to the mhra no later than the completion of the human pharmaco.